Background: Mantle cell lymphoma (MCL) accounts for approximately 5-7% of non-Hodgkin lymphomas (NHL). Bruton's tyrosine kinase (BTK) inhibitors, eg ibrutinib, are indicated for treatment of adults with MCL who have received ≥1 prior therapy. Primary and acquired resistance to ibrutinib is common and linked with poor outcomes, and remains an unmet medical need. Parsaclisib, a potent, highly-selective, next-generation PI3Kδ inhibitor, demonstrated clinical activity in patients (pts) with relapsed or refractory (R/R) NHL. We report preliminary results for parsaclisib monotherapy in a cohort of pts with R/R MCL who were previously treated with ibrutinib in the open-label, phase 2 study CITADEL-205 (NCT03235544).
Methods: Pts must be ≥18 years of age with pathologically confirmed MCL, R/R disease to the most recent treatment, documented cyclin D1 overexpression or t(11;14) translocation, have Eastern Cooperative Oncology Group performance status (ECOG PS) ≤2, and received 1 to 3 prior systemic treatments (including ibrutinib). Prior treatment with PI3Ki was prohibited. Pts were allocated to receive parsaclisib 20 mg once daily (QD) for 8 weeks followed by either 20 mg once weekly (weekly-dosing group [WG]) or 2.5 mg QD (daily-dosing group [DG]). Prophylaxis for Pneumocystis jirovecii pneumonia (PJP) was required. Objective response rate (ORR) was the primary endpoint; complete response rate (CRR), duration of response (DOR), progression-free survival (PFS), overall survival (OS), and safety and tolerability were secondary endpoints. All radiology-based endpoints were based on independent review.
Results: From October 2017 to January 17, 2020 (data cut-off), 47 pts were treated (WG, n = 12; DG, n = 35). Enrollment is ongoing. At cut-off, 70% of pts had discontinued treatment, most commonly due to progressive disease (62%). Median exposure (range) was 2.2 (0.1-18.0) months. Median age was 70 years and 79% of pts were men. Median time since initial diagnosis was 4.7 years. Most pts (85%) had ECOG PS ≤1 and 51% had high-risk MCL International Prognostic Index. The median number of prior systemic therapies was 3; 38% of pts had prior hematopoietic stem cell transplant, and 38% were refractory to most recent systemic therapy.
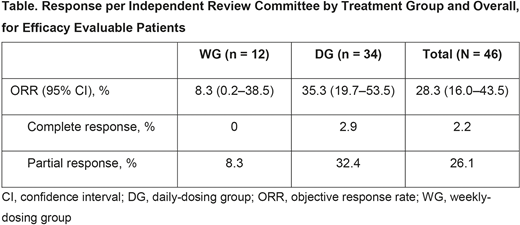
At data cut-off, 46 pts were evaluable for efficacy, including 12 in WG and 34 in DG (Table). Median follow-up (range) for this population was 10.2 months (1.5-25.9) overall and 7.6 months (1.5-25.9) for DG. ORR (95% confidence interval [CI]) and CRR were 28.3% (16.0-43.5) and 2.2%, respectively in all evaluable pts, and 35.3% (19.7-53.5) and 2.9%, respectively in DG. Median time to complete or partial response was 7.6 weeks. Median DOR (95% CI) was 7.3 months (0.2-not estimable) among all responders and 3.7 months (0.2-7.3) for DG. Median PFS (95% CI) was 3.65 months (1.9-3.9) overall and 3.65 months (1.9-5.5) for DG.
Among 47 safety-evaluable pts, most common treatment-emergent adverse events (TEAEs) occurring in >10% of pts were anemia (19.1%), diarrhea (19.1%), neutropenia (14.9%), asthenia and cough (12.8% each), decreased appetite, dyspnea, fatigue, pyrexia and rash (10.6% each). Most common grade ≥3 TEAEs reported in ≥5% of pts included anemia (12.8%), neutropenia (10.6%), thrombocytopenia and rash (6.4% each). TEAEs leading to dose interruption or reduction occurred in 31.9% and 2.1% of pts, respectively. TEAEs leading to treatment discontinuation occurred in 2 (4.3%) pts (diarrhea and colitis). Serious TEAEs reported in ≥2 pts were diarrhea, dyspnea, peripheral swelling and pneumonia (4.3% each). Two pts experienced fatal TEAEs (one pt had general physical health deterioration and respiratory tract infection, deemed not related to treatment; one pt had pneumonia, neutropenia, and septic shock, deemed related to treatment and disease progression). New or worsening grade ≥3 laboratory test values of clinical interest occurring in ≥5% of pts included decreased neutrophils (14.9%), platelets (10.6%), and hemoglobin (8.5%); there were no grade ≥3 increases in alanine/aspartate aminotransferase.
Conclusion: Preliminary efficacy data indicate that parsaclisib monotherapy is clinically active in this difficult-to-treat patient population. Treatment with parsaclisib had an acceptable safety profile and was generally well tolerated. Updated study results will be presented.
Zinzani:EUSA Pharma: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; AbbVie: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Gilead: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Kyowa Kirin: Consultancy, Speakers Bureau; Takeda: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; ADC Therapeutics: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Immune Design: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Janssen: Consultancy, Honoraria, Speakers Bureau; Sandoz: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Roche: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; BMS: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Servier: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Merck: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Portola: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Celltrion: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; MSD: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Verastem: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Incyte: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Immune Design: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Janssen-Cilag: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Kirin Kyowa: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; TG Therapeutics, Inc.: Honoraria, Speakers Bureau; Sanofi: Consultancy, Membership on an entity's Board of Directors or advisory committees; Eusapharma: Consultancy, Speakers Bureau. Delwail:Amgen: Consultancy. Paneesha:Bristol-Myers Squibb: Honoraria; Celgene: Honoraria; Gilead: Honoraria; Janssen: Honoraria; AbbVie: Honoraria. Rule:AstraZeneca: Consultancy; Celgene: Consultancy; Celltrion: Consultancy; Janssen Oncology: Consultancy, Research Funding, Speakers Bureau; Roche Pharma AG: Consultancy, Research Funding. Martin Garcia-Sancho:Roche, Celgene, Janssen, Servier, Gilead: Honoraria; Celgene, Eusa Pharma, Gilead, iQuone, Kyowa Kirin, Roche, Morphosys: Consultancy. Salles:Amgen: Honoraria; Bristol-Myers Squibb: Consultancy, Honoraria; Celgene: Consultancy, Honoraria, Research Funding; Gilead Sciences: Consultancy, Honoraria, Research Funding; Janssen: Consultancy, Honoraria; Novartis: Consultancy, Honoraria; Roche/Genentech: Consultancy, Honoraria, Other: Travel expenses, Research Funding; Servier: Consultancy, Honoraria; Acerta Pharma: Consultancy; Kite Pharma: Consultancy; Merck: Consultancy, Research Funding; Novimmune: Consultancy; Pfizer: Consultancy; Sanofi: Other. Sancho:Sandoz: Consultancy; Celltrion: Consultancy; Roche: Consultancy, Honoraria; Takeda: Honoraria; Novartis: Consultancy, Honoraria; Kern-Pharma: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; Gilead: Consultancy, Honoraria; Celgene: Consultancy, Honoraria; Bristol-Myers Squibb: Honoraria. Zheng:Incyte: Current Employment, Current equity holder in publicly-traded company. DeMarini:Incyte Corporation: Current Employment, Current equity holder in publicly-traded company. Jiang:Incyte Corporation: Current Employment, Current equity holder in publicly-traded company. Mehta:Seattle Genetics: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; TG Therapeutics: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Gelgene/BMS: Research Funding; Affimed: Research Funding; Merck: Research Funding; Kite/Gilead: Research Funding; Juno Parmaceuticals/BMS: Research Funding; Innate Pharmaceuticals: Research Funding; Oncotartis: Research Funding; Roche-Genentech: Research Funding; Incyte: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; fortyseven Inc/Gilead: Research Funding; Takeda: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.